What are carbohydrates?
Carbohydrates are starches, sugars and fibers found in vegetables, grains, fruits and milk products. They are one of the basic food groups and are generally considered important to a healthy life. Specifically, they are macronutrients which means they are one of the three main ways the human body obtains energy, called calories.
Carbohydrates get their name due to what they consist of at the atomic level. They are made up of carbon, hydrogen and oxygen atoms.
What is lactose?
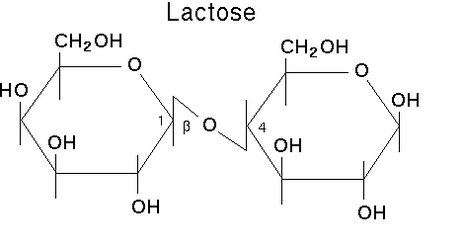
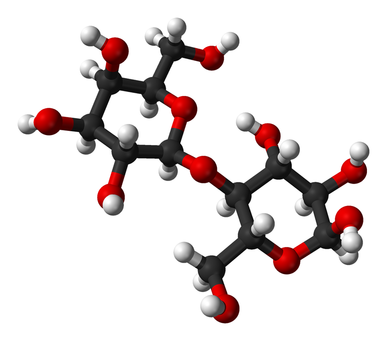
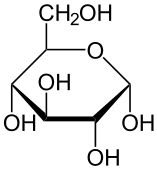
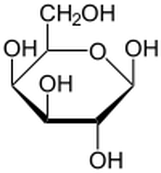
Lactose is a sugar present in dairy milk, specifically it is a disaccharide containing glucose and galactose units. It is an essential nutrient for the growth of infant mammals. Intestines of infants can secrete lactase, an enzyme that can help break down lactose, which is why they are very rarely lactose intolerant. As infants mature into adulthood, the production of lactase generally decreases due to their reduced milk consumption. Lactose supplies energy and if not digested in the upper intestine it contributes to developing a healthy bacterial system in the gut.
Structure of a lactose molecule
Carbohydrates are starches, sugars and fibers found in vegetables, grains, fruits and milk products. They are one of the basic food groups and are generally considered important to a healthy life. Specifically, they are macronutrients which means they are one of the three main ways the human body obtains energy, called calories.
Carbohydrates get their name due to what they consist of at the atomic level. They are made up of carbon, hydrogen and oxygen atoms.
What is lactose?
Lactose is a sugar present in dairy milk, specifically it is a disaccharide containing glucose and galactose units. It is an essential nutrient for the growth of infant mammals. Intestines of infants can secrete lactase, an enzyme that can help break down lactose, which is why they are very rarely lactose intolerant. As infants mature into adulthood, the production of lactase generally decreases due to their reduced milk consumption. Lactose supplies energy and if not digested in the upper intestine it contributes to developing a healthy bacterial system in the gut.
Structure of a lactose molecule
How does lactase break down lactose?
Lactase acts as the catalyst in the reaction that splits lactose into glucose and galactose. In metabolism, the glycosidic bond is hydrolyzed to form two molecular subunits. This reaction also produces heat. Chemically, the reaction is as follows:
C12H22O11 + H2O → C6H12O6 + C6H12O6 + Heat
Lactose + Water → Glucose + Galactose + Heat
In order for this reaction to happen, the optimum temperature is 37°C and the optimum pH is 6. The products of the reaction, glucose and galactose are simple sugars that can be absorbed through intestinal walls into the bloodstream.
Lactase acts as the catalyst in the reaction that splits lactose into glucose and galactose. In metabolism, the glycosidic bond is hydrolyzed to form two molecular subunits. This reaction also produces heat. Chemically, the reaction is as follows:
C12H22O11 + H2O → C6H12O6 + C6H12O6 + Heat
Lactose + Water → Glucose + Galactose + Heat
In order for this reaction to happen, the optimum temperature is 37°C and the optimum pH is 6. The products of the reaction, glucose and galactose are simple sugars that can be absorbed through intestinal walls into the bloodstream.
What is lactose intolerance?
Lactose intolerance is the inability to digest most of the lactose found in dairy products. This condition is caused by a shortage of the enzyme lactase which is used to break down the lactose into an absorbable form. Naturally, humans produce less lactase as they grow older, generally after two years of age, since the digestion of lactose is not necessary any more.
People who are lactose intolerant but drink dairy milk may feel very uncomfortable when digesting the milk. Common symptoms include nausea, cramps, bloating, gas, and diarrhea. These symptoms generally begin thirty minutes to two hours after drinking the milk.
There are various ways of diagnosing lactose intolerance some of which are entirely based on symptoms. One way of diagnosing the condition is for a person to fast for around two hours and then drink milk.Then, several blood samples are taken over the next two hours to measure the blood sugar (glucose) level. The blood sugar levels should rise if the lactose has been digested correctly otherwise they will stay the same meaning that the person is potentially lactose intolerant.
There is no treatment to improve the body’s ability to produce lactase. The best way to avoid the symptoms of lactose intolerance is to reduce the consumption of foods containing lactose. There are also lactase tablets/liquids available that allow people to digest lactose for certain periods of time.
Explanation of the chemical reactions used to isolate the lactose
Acetic Acid
10% acetic acid, in the form of vinegar, is added to the milk in order to separate the casein, a protein, within the milk so that it can be filtered out of the solution. The casein, present as calcium caseinate within the milk, has its isoelectric (neutral) point at a pH 4.6. This means it is insoluble in acidic solutions that have a pH less than 4.6. The pH of milk is generally about 6.6 which means the casein is soluble in milk. However, if acid is added to the milk, the negative charges on the outer surface of the casein micelle, the solubilized unit of casein, are neutralized (the phosphate groups are protonated). This causes the neutral protein, casein, to precipitate. The chemical reaction is as follows:
Ca2+Caseinate + 2HCl → Casein↓ + CaCl2
(Ca2+Caseinate is an ionic, colourless solution of calcium and casein, HCl is hydrogen chloride which is used as an acid and CaCl2 is calcium chloride, a salt, an ionic compound of calcium and chlorine)
This is why when vinegar is added to milk the casein present within the milk precipitates and it is possible to remove the casein from the milk (the casein↓ means it is a precipitate).
Calcium Carbonate
After the casein is removed from the milk, calcium carbonate, in the form of chalk, is added to the milk for two major purposes. The first of which is to neutralize the excess acetic acid that was added to the milk (calcium carbonate is basic with pH 9.4). Secondly, when the solution is heated to its boiling point, the initially soluble proteins of the albumin family (mainly lactalbumin and lactoglobulin) precipitate. Then, these proteins can be filtered out of the solution.
Ethanol
Lactose can be removed from milk (whey) by adding ethanol. Lactose is insoluble in ethanol which means when the ethanol is mixed with the aqueous solution, the lactose is forced to crystallize. The lactose crystals are then separated from the solution and their weight can be measured in order to find out the lactose content of the milk.
Lactose intolerance is the inability to digest most of the lactose found in dairy products. This condition is caused by a shortage of the enzyme lactase which is used to break down the lactose into an absorbable form. Naturally, humans produce less lactase as they grow older, generally after two years of age, since the digestion of lactose is not necessary any more.
People who are lactose intolerant but drink dairy milk may feel very uncomfortable when digesting the milk. Common symptoms include nausea, cramps, bloating, gas, and diarrhea. These symptoms generally begin thirty minutes to two hours after drinking the milk.
There are various ways of diagnosing lactose intolerance some of which are entirely based on symptoms. One way of diagnosing the condition is for a person to fast for around two hours and then drink milk.Then, several blood samples are taken over the next two hours to measure the blood sugar (glucose) level. The blood sugar levels should rise if the lactose has been digested correctly otherwise they will stay the same meaning that the person is potentially lactose intolerant.
There is no treatment to improve the body’s ability to produce lactase. The best way to avoid the symptoms of lactose intolerance is to reduce the consumption of foods containing lactose. There are also lactase tablets/liquids available that allow people to digest lactose for certain periods of time.
Explanation of the chemical reactions used to isolate the lactose
Acetic Acid
10% acetic acid, in the form of vinegar, is added to the milk in order to separate the casein, a protein, within the milk so that it can be filtered out of the solution. The casein, present as calcium caseinate within the milk, has its isoelectric (neutral) point at a pH 4.6. This means it is insoluble in acidic solutions that have a pH less than 4.6. The pH of milk is generally about 6.6 which means the casein is soluble in milk. However, if acid is added to the milk, the negative charges on the outer surface of the casein micelle, the solubilized unit of casein, are neutralized (the phosphate groups are protonated). This causes the neutral protein, casein, to precipitate. The chemical reaction is as follows:
Ca2+Caseinate + 2HCl → Casein↓ + CaCl2
(Ca2+Caseinate is an ionic, colourless solution of calcium and casein, HCl is hydrogen chloride which is used as an acid and CaCl2 is calcium chloride, a salt, an ionic compound of calcium and chlorine)
This is why when vinegar is added to milk the casein present within the milk precipitates and it is possible to remove the casein from the milk (the casein↓ means it is a precipitate).
Calcium Carbonate
After the casein is removed from the milk, calcium carbonate, in the form of chalk, is added to the milk for two major purposes. The first of which is to neutralize the excess acetic acid that was added to the milk (calcium carbonate is basic with pH 9.4). Secondly, when the solution is heated to its boiling point, the initially soluble proteins of the albumin family (mainly lactalbumin and lactoglobulin) precipitate. Then, these proteins can be filtered out of the solution.
Ethanol
Lactose can be removed from milk (whey) by adding ethanol. Lactose is insoluble in ethanol which means when the ethanol is mixed with the aqueous solution, the lactose is forced to crystallize. The lactose crystals are then separated from the solution and their weight can be measured in order to find out the lactose content of the milk.